Abstract
Bispecific antibodies are on the cusp of coming of age as therapeutics more than half a century after they were first described. Two bispecific antibodies, catumaxomab (Removab®, anti-EpCAM × anti-CD3) and blinatumomab (Blincyto®, anti-CD19 × anti-CD3) are approved for therapy, and >30 additional bispecific antibodies are currently in clinical development. Many of these investigational bispecific antibody drugs are designed to retarget T cells to kill tumor cells, whereas most others are intended to interact with two different disease mediators such as cell surface receptors, soluble ligands and other proteins. The modular architecture of antibodies has been exploited to create more than 60 different bispecific antibody formats. These formats vary in many ways including their molecular weight, number of antigen-binding sites, spatial relationship between different binding sites, valency for each antigen, ability to support secondary immune functions and pharmacokinetic half-life. These diverse formats provide great opportunity to tailor the design of bispecific antibodies to match the proposed mechanisms of action and the intended clinical application.
Via Krishan Maggon



 Your new post is loading...
Your new post is loading...





Highlights
•
Bispecific antibodies (BsAb) bind 2 distinct antigens or epitopes on the same antigen.
•
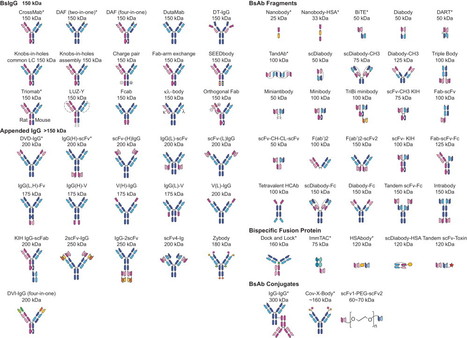
Antibody domain structure has been exploited to make >60 alternative BsAb formats.
•
2 BsAb are approved for therapy and >30 BsAb are in clinical development.
•
A common clinical application of BsAb is retargeting T cells to kill tumor cells.
•
Other clinical uses of BsAb include dual blockade of different disease mediators.
Molecular Immunology
Available online 27 January 2015
In Press, Corrected Proof — Note to users
Review Alternative molecular formats and therapeutic applications for bispecific antibodies ☆Christoph Spiess , Qianting Zhai , Paul J. Carter, Show moredoi:10.1016/j.molimm.2015.01.003Get rights and contentUnder a Creative Commons license